Do Nutrients Work Against Radiation?
If you or someone close to you has been diagnosed with cancer, then you are likely familiar with the most common treatment methods prescribed by traditional (western) medical practice. Chemotherapy, radiation, and surgery are used as a means to “kill” cancer cells or physically remove them from the body.
Many oncologists are not trained in nutrition and nutrient therapies and have a limited understanding of how these things work. Due to this lack of knowledge, they often have a bias against nutritional changes and nutrient therapies. They will often tell their patients that the nutrients will interfere with the radiation therapy they are using. This article will address whether nutrients work against radiation.
What is Radiation Therapy?
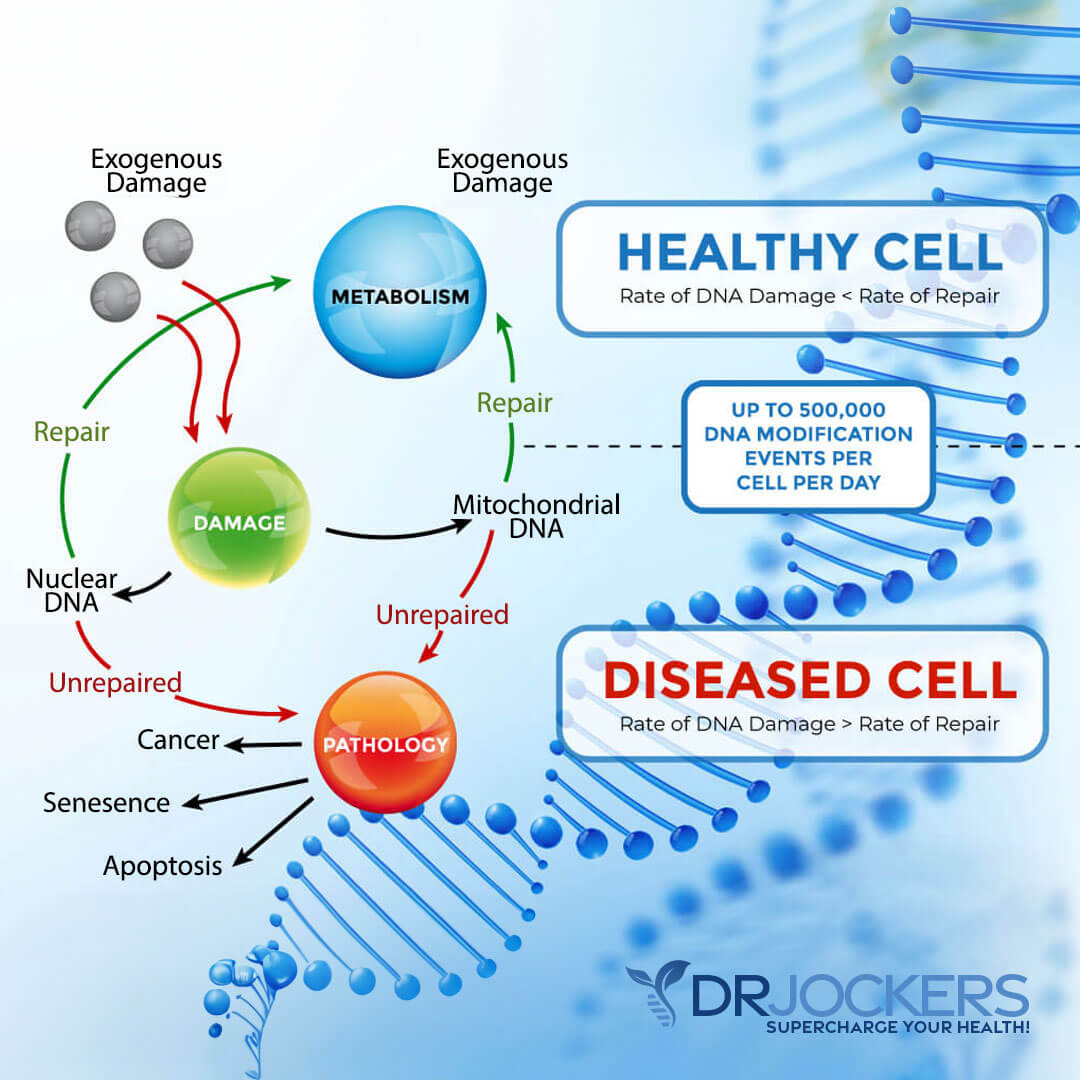
Radiation is a type of energy found to be cytotoxic, meaning poisonous to cells. We are exposed to radiation every day from the sun, our cell phones, and x-rays. Even low levels of radiation can accumulate in the body over time and it has been observed that this significantly contributes to the formation of free radicals.
High concentrations of free radicals in cancer tissue are used to create damage to cancer cell DNA. The goal is to disrupt the DNA of the cancer cell enough that it will be unable to continue dividing and die because the DNA contains the instructions of the cell.

Nutrition and Radiation Therapy
As doctors are aware, however, radiation can also damage healthy cell DNA and even promote cancer formation. This is because free radicals work by pulling electrons off of molecules and making them unstable which is what leads to the desired effect of damaging cancer cell DNA.
The downside to this is that these free radicals have the same potential to affect healthy tissues and, depending on the site in the body, can interrupt ordinary cellular function.
Fortunately, the administration of specific nutrient therapies can protect healthy cells from the oxidative (damaging) effects of the radiation-induced free radical formation while making cancer cells more vulnerable and therefore easier to kill.
Specific nutrient therapies will not only assist in ridding the body of cancer but will also mitigate several harmful side effects of radiation treatment, improve patient well-being and preventing long-term complications.
Radiation and Antioxidants
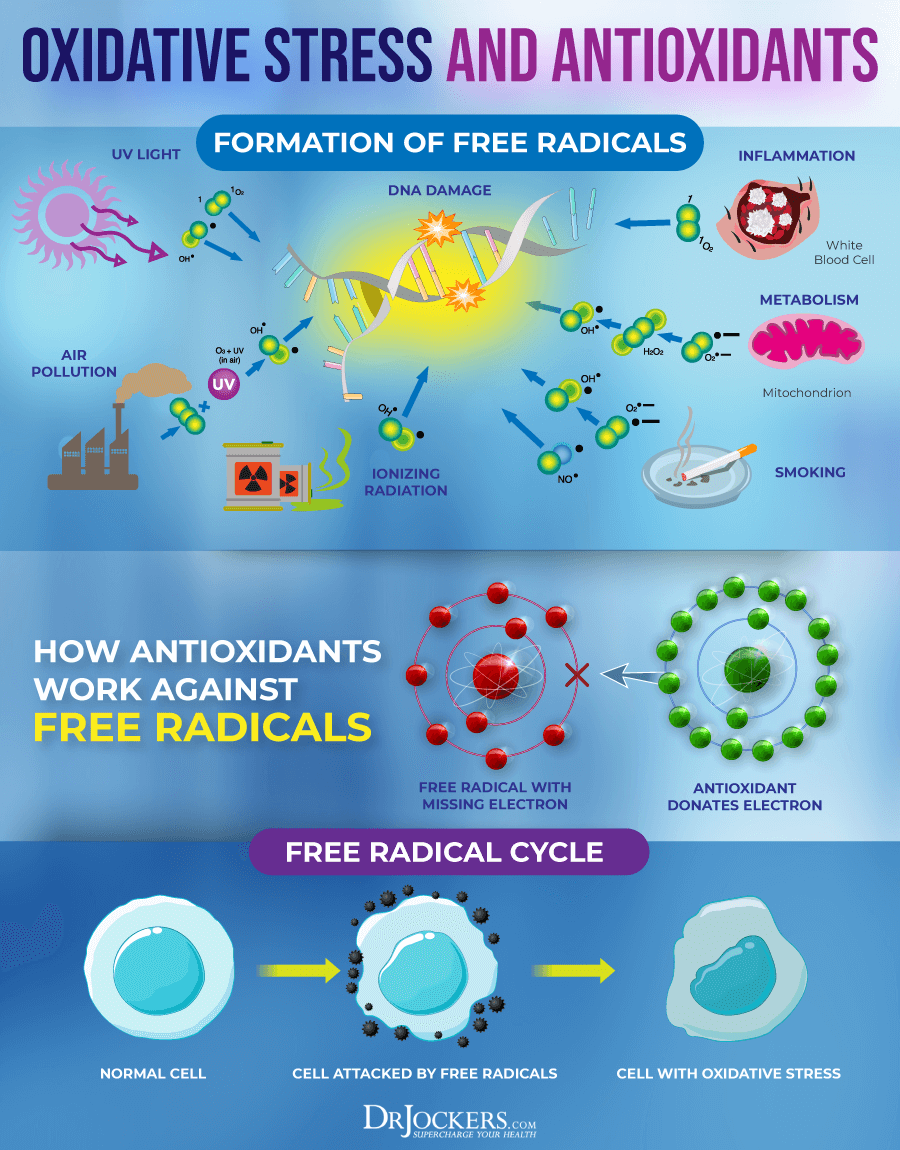
Antioxidant use in conjunction with radiation therapy has long been a topic of controversy and (predating current scientific evidence) there were concerns that they could potentially interfere with treatment.
Combining the oxidative effects of radiotherapy with an antioxidant therapy may seem like a bit of a self-negation. This was a long-held conventional theory. More recent research, however, has been conducted on this specific concern and the results provide a paradigm-shifting conclusion.
Not only do dietary antioxidants not interfere with radiation therapy, but they actually make radiotherapy more effective and less harmful to healthy tissues (1).
Among the antioxidants reviewed were vitamin A, vitamin E, selenium, vitamin D, glutathione, the B vitamins, curcumin, and cysteine.
Importance of Glutathione
An important strategy for protecting and repairing DNA is boosting systemic glutathione levels. Glutathione has been dubbed the “master antioxidant” and for good reason. Not only does it have free radical mitigating properties on its own, but it also helps regulate other antioxidants in the body such as vitamin C and E.
High levels of glutathione have demonstrated a remarkable ability to control oxidative stress by neutralizing free radicals, particularly in the case of radiation exposure (17). The master antioxidant can be boosted naturally by consuming foods that contain the precursors glycine, glutamic acid, and cysteine.
The best sources of these amino acids are a 100% grass-fed un-denatured whey protein or a high-quality pea protein. NAC (N-acetyl cysteine) and ALA (alpha lipoic acid) supplementation are also able to raise glutathione levels. Finally, for optimized glutathione levels it is important to include flavonoid rich foods and adequate magnesium in the diet.

Cancer and Patient Micronutrient Status
Cancer patients are typically deficient in critical micronutrients before treatment and become further malnourished throughout treatment (2, 3). Additionally, even slight malnutrition leading into treatment will increase the chances of suffering side effects from radiation damage.
This effect is even greater when radiation and chemotherapy are combined. Increasing the intake of several natural compounds derived from food and plant sources can have a powerful protective effect against these damages.
It is important to consider that radiation has a statistically significant tendency to be reflected off of certain tissues in the body (such as the bones) and other tissues. This phenomenon is referred to as scatter radiation.
When receiving a radiation treatment, it can be hard to determine which areas of the body are being damaged due to scatter radiation results. It is important then to strengthen the whole-body protection process by improving the overall micronutrient status of the body.
This can be done by incorporating a wide range of organic vegetables that are rich in cancer protective compounds such as flavonoids and carotenoids. Increasing intake of these compounds increases the body’s own ability to fight cancer while also providing natural defenses against many forms of radiation.
Protecting Healthy Cells
To prevent healthy cell damage and promote healing in healthy cells that have already been damaged, it is important to concentrate on DNA repair enzymes. A high intake of antioxidants has been shown to protect the integrity of DNA repair enzymes while consumption of L-carnitine, zinc, and the b-vitamins 6, 12, folate, and niacinamide has shown to increase the number of repair enzymes in some cases (4, 5).
A quercetin and vitamin C combination also has powerful DNA-protecting effects. These nutrients do not have the same protective effect over cancer cells, meaning they protect healthy cells and allow cancer cells to die when exposed to radiation.
Another Important approach to protect healthy cells is to limit inflammation as much as possible. In addition to an antioxidant-rich diet, inflammation can be controlled effectively by optimizing vitamin D levels, consumption of high-quality omega-3 fatty acids, and dietary inclusion of turmeric or curcumin (6, 7, 8).
Another powerful strategy for limiting inflammation in the body is cutting sugar and grains from the diet and opting for a ketogenic style eating plan. This style of eating emphasizes the intake of healthy fats and a very low intake of carbohydrates to promote the body to burn fat as its primary fuel source.
Eating a ketogenic style diet will normalize insulin levels, help control inflammation and can sensitize cancer cells to radiation therapy (9).
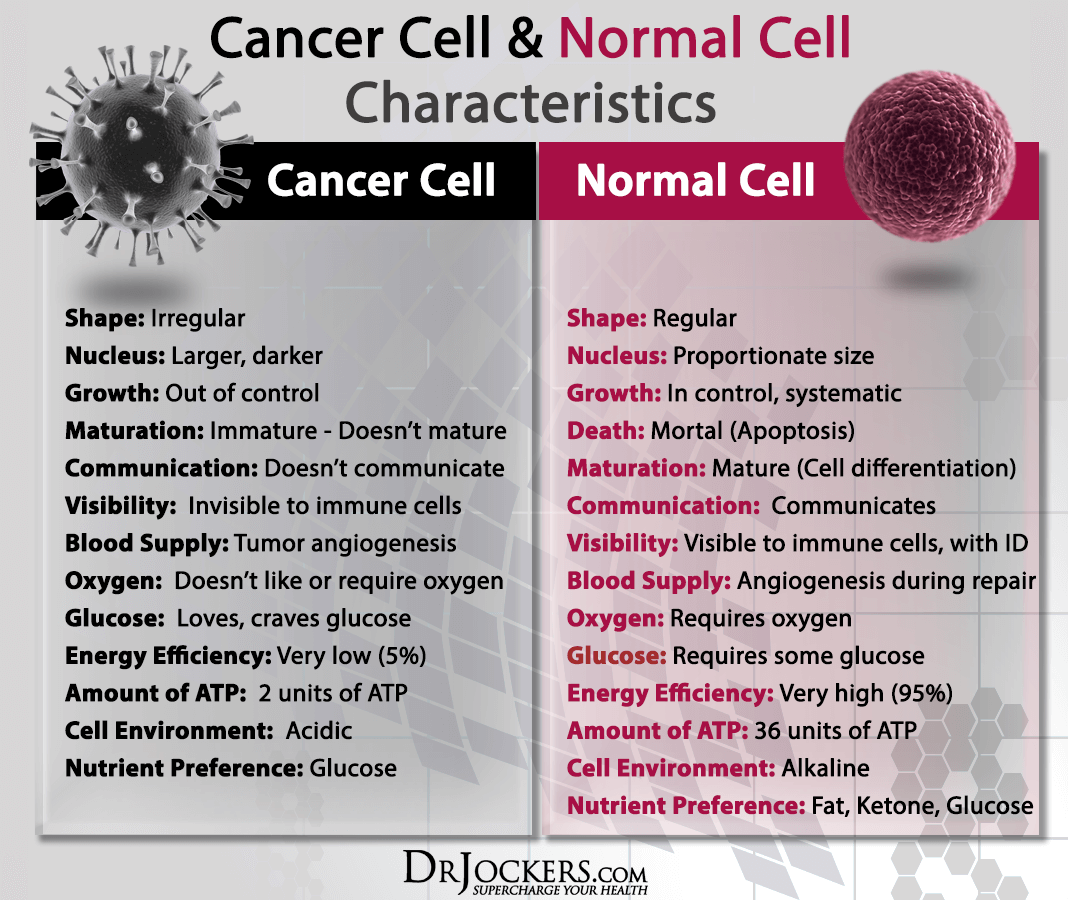
Damage to Healthy Cells
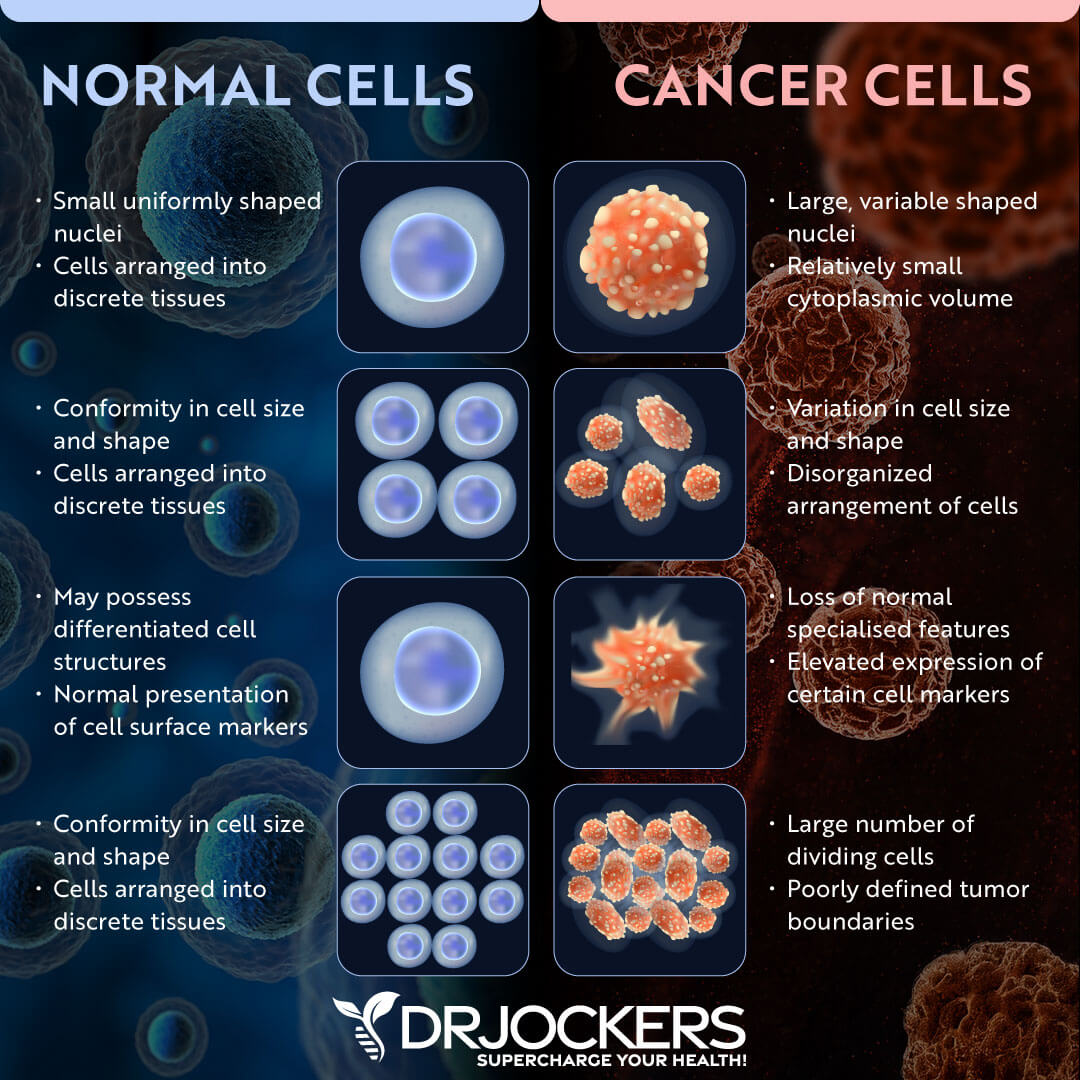
The most accepted and widely understood side effect of radiation therapy is that it can damage healthy cells in addition to cancer cells. This damage can cause a list of side effects depending on what part of the body has been irradiated.
For example, irradiation of the spine or brain can lead to radionecrosis, a rubbery growth along the nervous system that can lead to neurological complications. Even in the face of more advanced and targeted radiation administration strategies, it has been a great challenge in western oncology to protect non-cancerous cells from the damage of radiation.
Radiation accumulates in the tissues and generates large amounts of free radicals, even after treatment has been stopped. To mitigate acute and delayed damage to normal cells during radiotherapy, several natural compounds should be considered and ideally, the patient should begin consuming them before treatment has begun.
Protecting the Nervous System
Damage to the nervous system during chemotherapy or radiation treatment is a major concern. Often patients report feeling mentally inhibited shortly after beginning radiation treatment and can persist several years after treatment has been completed.
It has been demonstrated clinically that radiation can cause damage to the brain and other structures of the nervous system even when not in the direct path of the beam. Radiation of the hand has even been shown to promote oxidative stress and inflammation in the brain (10).
Another side effect of radiation therapy that causes concern is the aforementioned radionecrosis. Unlike the immediate presence of mental sluggishness, radionecrosis usually does not develop until several years after treatment and is associated with its own line of neurological impairments.
Radionecrosis and other presenting nervous system damages have been shown to respond well to the addition of certain nutrients to the treatment. For the purpose of protecting the nervous system, a combination of the following can be used: mixed carotenoids, magnesium, vitamin E succinate, green tea extract, quercetin, curcumin, ALA, and Omega-3.
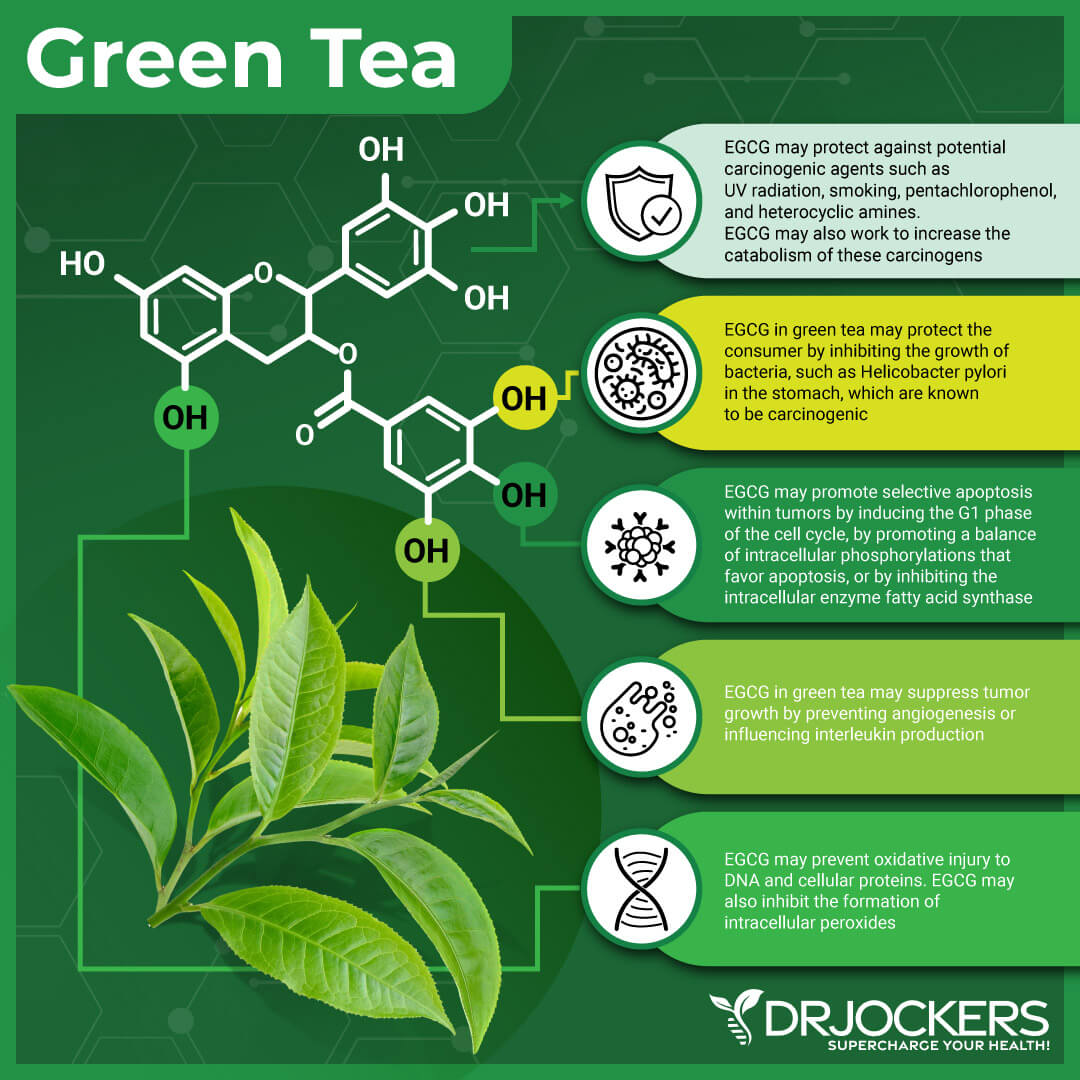
Hyperbaric Oxygen
Another effective intervention for protecting the nervous system is hyperbaric oxygen therapy (HBOT). HBOT is when a patient is placed in a chamber that concentrates oxygen at a pressure greater than that of the normal atmosphere and promotes a measurable increase in oxygen levels in the body.
A recent study reviewed the literature concerning HBOT in combination with chemo and radiotherapies and concluded that it did not interfere with normal treatment and in many cases significantly improved several measures of treatment.
Applied during radiotherapy treatment, HBOT demonstrated the ability to sensitize cancer cells to radiation while exerting protective effects over healthy cells. This effect was especially prominent in malignant gliomas in which radiation is often applied directly to the spine or brain (11).
Another review study looked at over 2500 patients with delayed onset radiation injuries who received HBOT and, of those with forms of radionecrosis, improvement in symptoms ranged from 76.7-92% (12).

Protecting the Digestive Tract
As with other rapidly dividing cells, the cells of the digestive tract are particularly prone to damage by radiation treatment. As we now know, the health of the gut can have a significant impact on one’s overall well-being.
The body relies unrelentingly on a healthy digestive tract to properly digest food, regulate the immune system, and help block dangerous toxins from entering the bloodstream. To maximize long-term health outcomes of radiation or chemo/radiation therapies, it is important to protect the health of the digestive tract – more so if nutrient therapies are going to be used.
The first major issue with radiation and the gut is that it destroys the biodiversity of the microbiome (gut bacteria) and allows harmful microbes, such as H Pylori and candida to flourish. When harmful bacteria overgrow the beneficial bacteria in the gut, it is called dysbiosis and these bad microbes increase gut inflammation and are able to enter the bloodstream.
This is a problem because they have the ability to lower secretory IgA, a critical immune system regulating factor that is highly concentrated in mucous membranes. They also release toxins that promote inflammation and sometimes lead to autoimmune reactions.
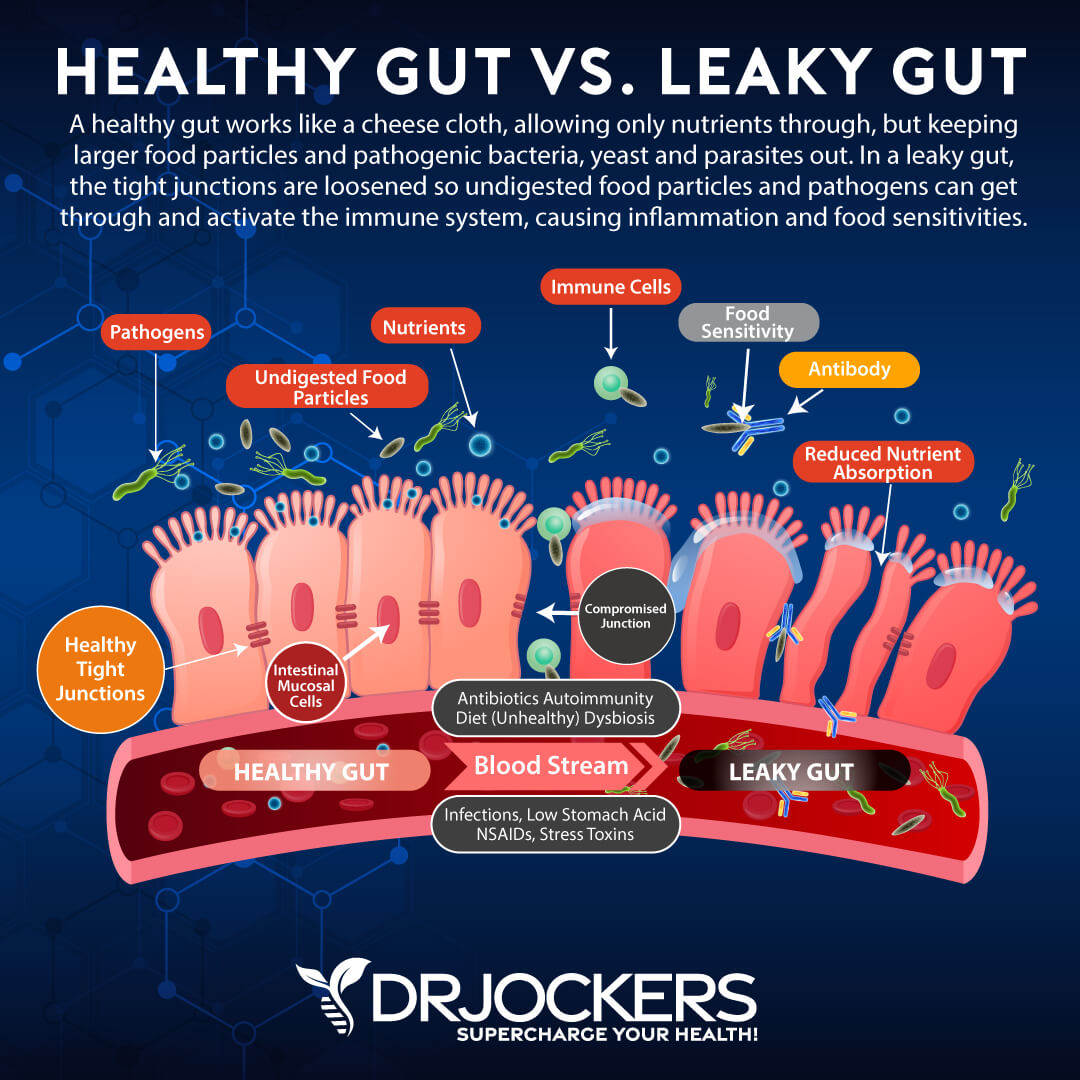
The Development of Leaky Gut
Another area of concern is the general damage to the cells of the digestive tract lining. Consequences of this damage are lowered mucosal secretions, severe inflammation of the digestive tract, inhibited digestion leading to malnourishment, and loosened gap junctions between cell membranes leading to leaky gut.
Combined, these damages can lead to detracted immunity, lowered cancer-healing efficacy, and the inevitable discomforting side effects. Fortunately, many of the nutrients that protect the nervous system from radiation injury also provide protection for the gut, particularly curcumin and quercetin.
A recent review of the literature established the importance of bacterial diversity in the gut, especially during cancer treatment, as it may exert powerful effects on mucosal secretions and intestinal repair mechanisms (13).
Consuming a variety of fermented foods, such as kimchi and sauerkraut, as well as a broad-spectrum probiotic can provide this therapeutic effect. In addition, nutrients such as green tea extract, ginger, ellagic acid, zinc, bioflavonoids, L-glutamine, MSM, and selenium along with prebiotic nutrients such as inulin are especially helpful in protecting and repairing the gut lining. I recommend my clients who are undergoing chemotherapy or radiation use Gut-Liver Cleanse Protein to protect and heal the gut lining.
Alkaline Phosphatase
Another potential mechanism to protect the gut from radiation injury is through the upregulation of the enzyme alkaline phosphatase. Alkaline phosphatase is present throughout the body and has key roles in the gut to control inflammation, regulate the microbiotic diversity of the gut lining, and strengthen the gap junctions between gastrointestinal cells (14).
It also helps neutralize endotoxins in the gut which can substantially lower the toxic load on the liver and kidneys and prevent diversion of the immune system from fighting cancer. Endotoxins can also be responsible for activating inflammatory processes so by neutralizing them, systemic inflammation will also be lowered.
An animal study demonstrated in rats with radiation-induced GI tract damage and lowered alkaline phosphatase that administration of exogenous alkaline phosphatase to the intestines significantly mitigated inflammation and helped restore bacterial diversity (15).
The turmeric compound curcumin has demonstrated the ability to naturally boost low intestinal alkaline phosphatase levels suggesting it may be of therapeutic benefit for radiation-induced GI damage (16).
It may be advantageous to test alkaline phosphatase levels before beginning treatment as low levels may be caused by a zinc deficiency. We offer an indepth test, called the Comprehensive Blood Analysis which includes alkaline phosphatase and the rest of the key liver enzymes as well as a complete blood count to look at immune factors, blood cell factors, signs of leaky gut and low stomach acid, gallbladder health, vitamin D status, thyroid and inflammatory levels in the body. You can find this test here.
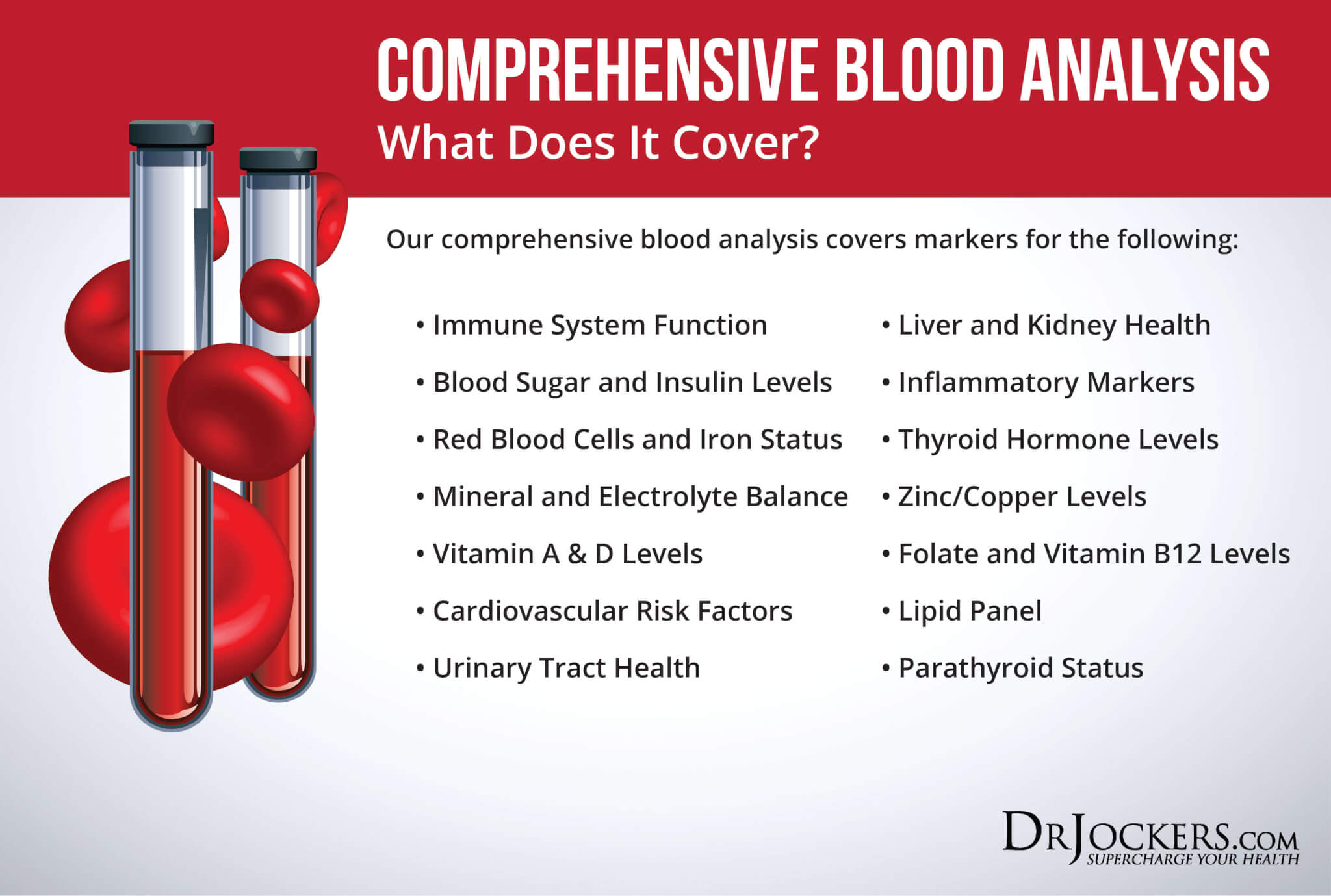
Enhancing Radiation Therapy Outcomes
Several natural compounds are known to act on specific mechanisms of cancer formation to limit its growth and promote apoptosis.
Many of these nutrients can improve the effectiveness of radiation when administered before, during, and after treatment by inhibiting cancer cell growth, protecting healthy cells, and boosting the immune system.
Inhibiting Cancer Cell Growth
It is known that sometimes radiation can irritate cancer cells and cause them to respond more aggressively. The response to this is often to also increase the aggressiveness of the radiation treatment, often leading to more severe side effects due to systemic damage to the body.
Nutrients that act to promote cancer cell dormancy and lower systemic inflammation can be incorporated into radiotherapy treatment to prevent cancer cell irritation and lower the radiation dose required to kill the cancer. A combination of mixed carotenoids with niacinamide can be introduced in order to promote cancer cell dormancy and increase the effectiveness of radiation for killing cancer cells.
Not unlike the reasoning behind the aforementioned HBOT protocol, increasing the oxygen content of a tumor will dramatically raise its vulnerability to radiation, which occurs with niacinamide supplementation.
Another strategy that has drawn interest and focus is the inhibition of the inflammatory COX enzymes. Drugs administered that act to lower COX enzymes effectively slow cancer growth and natural substances such as resveratrol, curcumin, and quercetin can provide the same effect in a safer and more effective manner.

Key Immunity Boosting Compounds
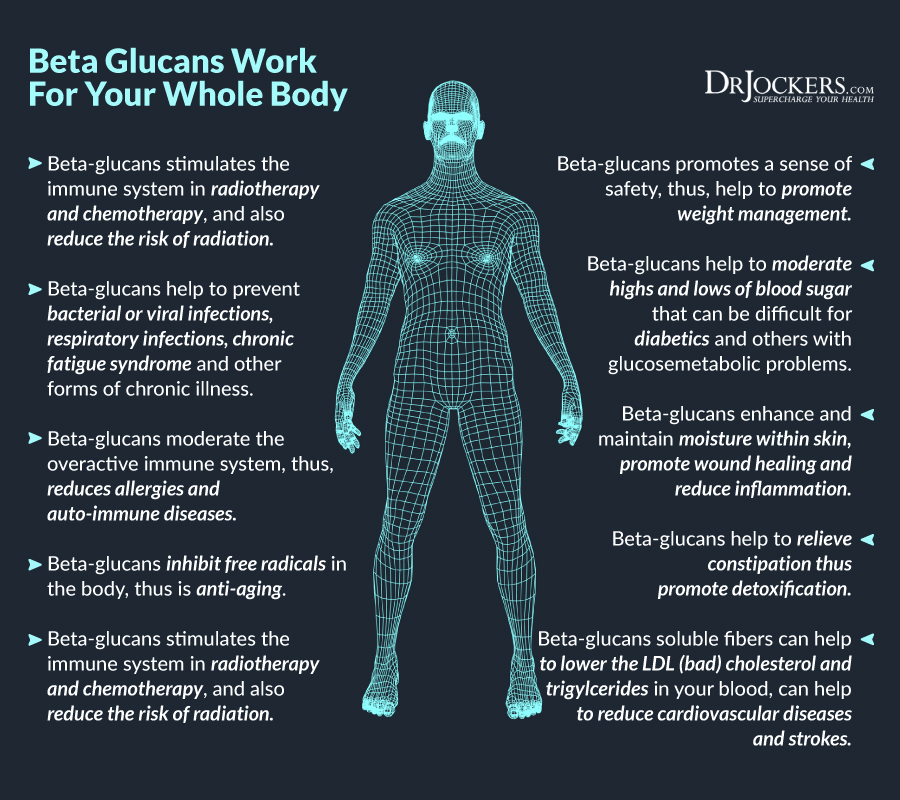
Radiation puts a high amount of stress on the body. High levels of stress tend to inhibit the immune system and this is problematic for someone trying to fight off cancer. In addition to many of the compounds already mentioned, such as flavonoids and carotenoids, beta glucans have powerful immune boosting effects.
Specifically, beta-1,3-glucan has been studied for its ability to restore immunity when it has been compromised, such as with radiation treatment. Additionally, Beta-1,3-glucan has an anti-cancer influence as an immune system modulator because it interacts with natural killer cells to tag cancer cells for destruction (18).
What is significant about beta-1,3-glucan, and why it can be critical in cancer treatment, is that it is an immunomodulatory compound. This means that it doesn’t just boost immunity, but it also prevents the over-stimulation of the immune system and may help control auto-immune reactions.
Final Thoughts
The strategies listed above are all ways to make the task of enduring radiation therapy easier to endure while boosting one’s long-term health. Implementing nutrition-based therapies alongside conventional cancer treatment is not only helpful but is crucial to maximize one’s body’s cancer fighting abilities.
Oncological radiologists usually do not receive education on natural compounds so consulting a natural health professional who has this knowledge will provide valuable insight.
If you want to work with a functional health coach, I recommend this article with tips on how to find a great coach. Our website offers long-distance functional health coaching programs with our world-class team of health coaches. For further support with your health and other goals, just reach out—our fantastic coaches are here to support your journey.

Inflammation Crushing Ebundle
The Inflammation Crushing Ebundle is designed to help you improve your brain, liver, immune system and discover the healing strategies, foods and recipes to burn fat, reduce inflammation and thrive in life!
As a doctor of natural medicine, I have spent the past 20 years studying the best healing strategies and worked with hundreds of coaching clients, helping them overcome chronic health conditions and optimize their overall health.
In our Inflammation Crushing Ebundle, I have put together my very best strategies to reduce inflammation and optimize your healing potential. Take a look at what you will get inside these valuable guides below!








This is so true, our father in-law refused to listen to our recommendations on how he could relieve his nausea using food. His doctor told him the same, as if food were the enemy and medicine the savior.
Yes, this is all too common in the medical model and that is why my team and I are putting together articles like this and creating a book full of research studies on nutrient therapy alone or in combination with conventional treatments for cancer.
Thank you Dr. Jockers. This is exactly what I was looking for. I am starting radiation with low dose chemo after 2 1/2 years of natural treatment. No mestasis but my tumor has grown alot, is extremely painful and bleeding which gave me severe anemia and needing blood transfusions. How do I work with my oncologist on this? She told me to only take a multi vitamin. Thank you so much for your work. You feel like a friend from watching your videos.
So sorry to hear about this! I would encourage you to work with both your oncologist and a functional health practitioner to customize a good program. Here is a helpful article: https://drjockers.com/functional-nutrition-tips-to-find-a-great-health-coach/