The Science Behind Magnesium Stearate
There are so many health products and supplements available that it makes it quite confusing for the average consumer to figure out what they should get. As a clinician, I see so many of my patients bringing in bags of cheap and ineffective supplements. These are made with cheap nutrient sources and potentially dangerous filling agents.
I have seen many individuals develop digestive problems and other health issues that I believe were at least partially due to the overconsumption of filling agents in various supplements. As a clinician, I have always valued the highest level of purity in the products I recommend to my clients.
In the process of formulating my own supplements, I was able to work with some research biochemists and was enlightened on the use of one major ingredient – Magnesium Stearate (MS). Research shows that MS is benign, yet due to several websites, has developed a bad rap.
I was avid about not using this product until I was challenged to actually study it for myself. Once I dug into the literature, I realized I had made a blind assumption that this was a bad product without really understanding what it was.
Here are the results of my indepth research into the health controversy behind magnesium stearate.

What is Magnesium Stearate?
Magnesium stearate is a salt that is formed by a magnesium ion that bonds with two stearate molecules. A salt has a cation (+ charge) and an anion (- charge). Magnesium is the cation and stearic acid is the anion.
Stearic acid is a long-chain saturated fat that is found most commonly in beef, cocoa butter, coconut oil, palm oil and other meats and vegetables. Studies have shown that stearic acid does not raise cholesterol levels or increase the risk of heart disease (1, 2, 3).
Some studies have shown that stearate has health benefits in that it does not elevate post-meal blood lipids or increase clotting factors (4). This is a normal fatty acid that we all consume on a regular basis that has potentially beneficial effects on the body. Magnesium is a key mineral that plays a role in an enormous amount of reactions within our body.
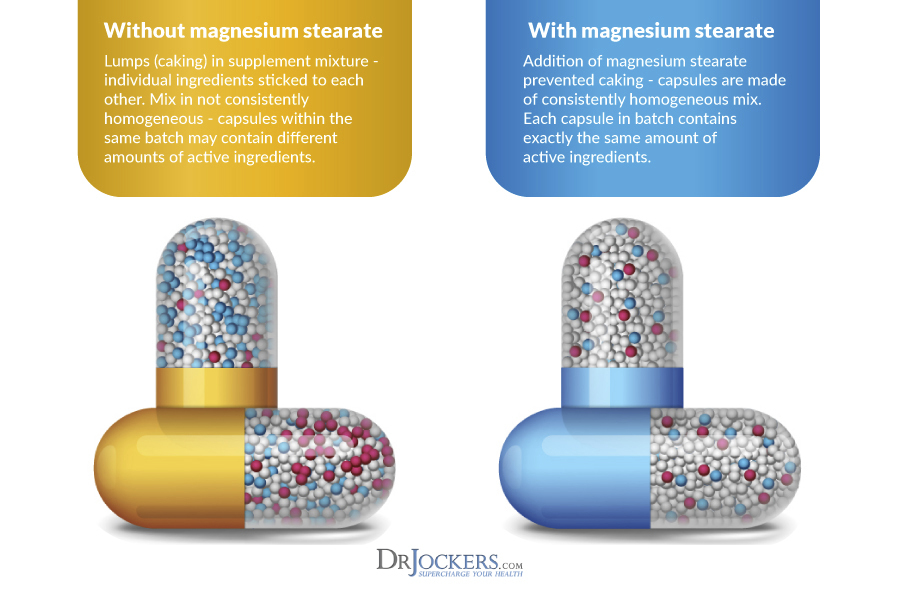
Why Do Manufactures Use Stearates:
Many supplements use stearic acid, vegetable stearate and/or magnesium stearate. These substances have lubricating properties and prevent ingredients from sticking to manufacturing equipment during the compression of chemical powders into capsules or tablets.
Most pharmaceutical grade (highest level of purity) formulations include a certain amount of lubricant to improve their flowability and prevent their adhesion to the surfaces of processing equipment.
Without the usage of flow agents, it is hard to ensure consistent dosages of the active ingredients in each supplement. These flow agents are considered by the FDA to be extremely important for quality control of the supplement.

What We Know About Magnesium Stearate:
- Stearic Acid and stearates are found in many natural foods we eat every day
- Stearates have been shown to have a positive effect on blood lipids and clotting factors
- Magnesium is a key mineral used in thousands of biochemical processes in our body
- Magnesium stearate is a benign ingredient that improves the flowability of supplement powders as they go through processing equipment.
- Stearates are considered normal and important parts of the pharmaceutical grade formulation process.
Claims About Magnesium Stearate:
There are many claims around the internet about magnesium and other stearates causing various problems in the body including:
- Biofilms in the Digestive System
- GMO’s and Pesticides
- Reduced Nutrient Absorption
- Reduced Immune Function
- Allergy Risk
I will do my best here to address those issues here in this article.
Intestinal BioFilms:
An intestinal biofilm is a layer of slime released by intestinal microbes to protect themselves from the immune cells of the body. Intestinal biofilms can be a major health threat because they can protect pathogenic microbes from natural immune regulation.
Some of the critics of magnesium stearate believe that it can induce the formation of similar biofilms in the intestines. This is based around the chemical makeup of soap scum with contains magnesium and calcium stearate. The claim is that soap scum creates films on the sink and shower and thus, the magnesium stearate will create a similar film on the intestines.
This is a mistake of association and not causation. There are many different ingredients in soap that contribute to the soap scum formation. To blame it on the stearates is not scientific. Additionally, our intestines are a much different environment than a sink or shower. Finally, there is no scientific data that suggests this is a possibility.
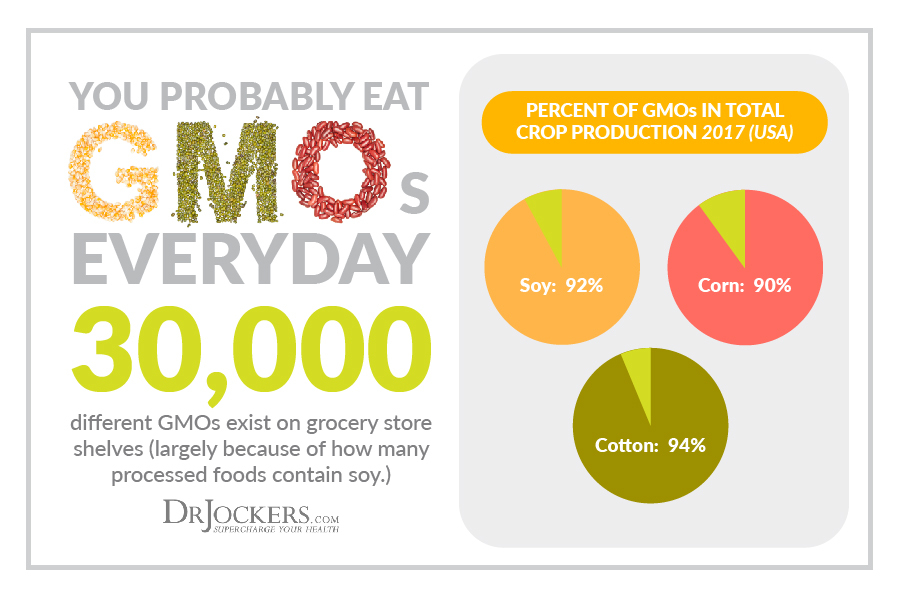
Cottonseed Oil and GMO’s:
Many stearates are derived from cottonseed oil which is genetically modified (GMO). GMO’s are full of dangerous pesticide chemicals. This is a substantial concern in that pesticides can cause substantial damage to the body and we want to be sure to avoid GMO’s and pesticide content in our food and supplements.
I am always looking for companies to have certain labels such as Non-GMO verified and/or statements saying that the product does not contain genetically modified ingredients.
We all should be aware that magnesium stearate is a highly purified substance that goes through a thorough refining process before it is used in supplements. I have yet to find any research linking magnesium stearate and pesticide risk.
Nutrient Absorption:
Critics believe that magnesium stearate may inhibit nutrient absorption through the digestive system. This is based on a 2007 study where tablets containing magnesium stearate dissolved slower than tablets without magnesium stearate in an artificial gastric juice (5).
The study authors were inconclusive in their report saying that further in vivo studies were needed to determine what sort of practical significance this finding posed. An earlier, 1998 study found no effect on tablet dissolution at all (6).
A 1997 study found that although magnesium stearate increased dissolving time, it had no effect the bioavailability of the drug used in the study. The effects of the drug were similar in both the magnesium stearate group and the control group as seen in the blood levels of the patients (7).
So based on these studies, we could conclude that magnesium stearate may or may not affect the dissolving time but does not affect the overall effect of the drug or supplement.

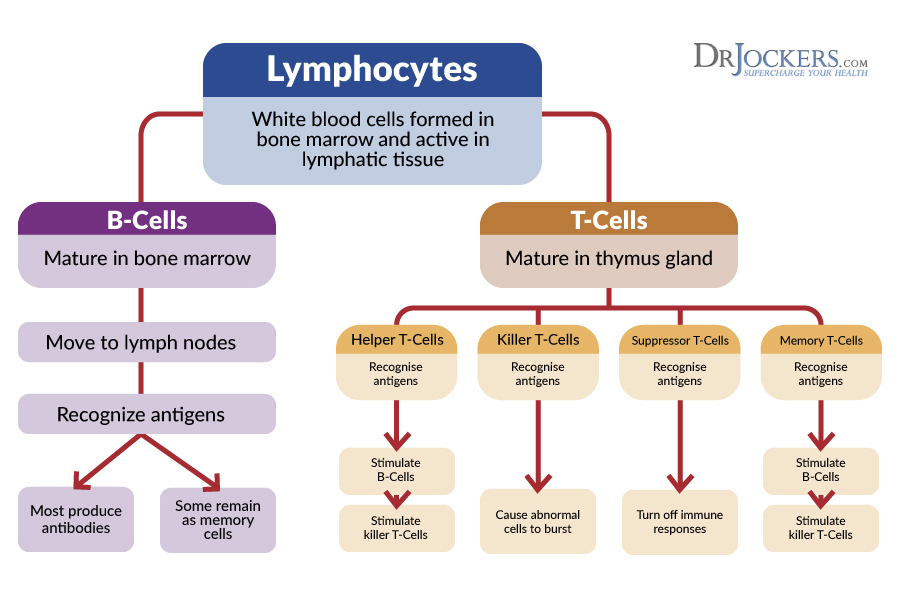
Immune System Function:
Some critics believe that magnesium stearate may have a harmful effect on the immune system. This is based around one major study done in 1990 found that stearic acid may have negative effects on the immune system (8).
In the experiment, scientists took immune cells and saturated them with stearic acid based solution. They observed that the T-cells incorporated enough stearate into their cell membranes that the membranes became destabilized and the cells died.
This study should not be used to form real life assumptions. Under normal conditions, our T-cells will never be bathed in stearic acid, no matter how much chocolate, coconut oil, cocoa butter or magnesium stearate we consume.
The researchers also used T-cells from mice, and therefore the results cannot always be applied to humans. As the study authors discussed, the mouse cells incorporated stearic acid into their membranes because they lacked the ability to de-saturate fatty acids.
Studies have shown that human T-cells do have the ability to de-saturate fatty acids (9) so the results of the former study would most likely not take place. Based on current evidence we should not be concerned about magnesium stearates and stearates in general inhibiting healthy immune function.
Allergy Risk:
A case study published in 2012 reported a 28-year old woman who had a severe allergic reaction to magnesium stearate. The woman broke out in hives due to an alleged allergy to the substance (10).
There is very little surrounding evidence that would make one suspect magnesium or stearate as potential allergens but unique and rare cases are plausible. Just the same, people have very rare allergic reactions to nearly every food and herb on the planet.
If you develop hives or any other allergic like response after consuming magnesium stearate, you should obviously discontinue usage immediately.
A short-term safety study was performed in 1980 to see the highest levels of tolerance of magnesium stearate. The study used rats and found that 2500 mg per kilogram of bodyweight per day was necessary to begin seeing toxic effects (11).
This result is equivalent to a 150 lb person consuming 170,000 mg per day, which is far beyond what you would consume even if you were taking 100 capsules a day. The average capsule contains less than 10mg of magnesium stearate.
Conclusion:
There is not enough scientific data to claim that magnesium stearate poses any possible risks. I believe that those who are making big headline articles about the dangers of magnesium stearate are doing it to market their products as if they contain a higher level of purity.
I would be sure to stay away from products that do not have a statement about not including any ingredients that are genetically modified. Additionally, I would stay away from products containing artificial colorings, flavorings or preservatives. Other ingredients to avoid would include titanium dioxide, parabens, MSG, carrageenan, hydrogenated oils and sodium benzoate.
DrJockers.com Stance on Stearates:
After reviewing the literature and having discussions with product manufactures and nutritional biochemists, I changed my stance on magnesium stearate.
We have chosen to use magnesium stearate in many of my products on DrJockers.com because of the safety and the thorough focus we have on making sure our products are the highest grade possible.
We are also completely transparent about the ingredients in the supplement as opposed to many companies who do not reveal the binding ingredients they use (as manufactures are not enforced to list hypoallergenic ingredients).
We want you to understand that the decisions we make at DrJockers.com are after much diligence, research, thought and prayer. We are not perfect, but we are humble enough to change our opinions when enough evidence is given to us. This is such an example.
We appreciate your attention and business and strive to serve you all at the highest level.






I developed hives after taking a supplement (alpha lipoic acid) that contained the filler magnesium stearate. I got a different brand that did not have this filler (Kirkmans) and did not get hives. However, after a while the powder supplement in Kirkmans started clumping and sticking together. I was still much better than getting hives.
Thanks for sharing Helena, this is extremely uncommon. Glad you found something that worked for you!
I will be going to get this allergy test. Magnesium stearate is in 2 very different supplements that I have taken, causing the same trouble itchy rash allergic reaction. Kirkland brand (from Costco) Fast Acting Lactase, and AdvoCare brand “ThermoPlus”.
I was told that Magnesium stearate was good for sleep at a local health food store, is this wrong? Did I buy the wrong Magnesium?
Hey Hector, magnesium stearate isn’t the best for sleep. I like Magnesium L-threonate for that. Check this article out: https://drjockers.com/improve-sleep/
Good Morning, can you please tell me if your products are made in the USA? Thank you.
Yes they are Christi!
Dr Jockers,
Your article was very Interesting. I have become either sensitive or allergic to magnesium stearate. I have been having migraines since the mid 1980’s. My wife and I have gotten more and more concerned since the headaches last as long as three (3) at a time. Several of my medications have MS in them and we wondered of it was the common denominator for the migraines.
We were coming home after visiting friends.
We stopped on the interstate to stretch and use the facilities. After doing so we bought bottled water and headed back to the car. Prior to pulling into traffic I opened my bottle and took a drink. After doing this we got back on the interstate to do the last leg of our journey but within 2-3 minutes I was blessed with a whopper of a migraine. I told my wife and she looked on the label for the water and found that it had MS.
After getting home I spent the next three days in bed with a raging migraine. I know without a doubt that MS is a trigger for my migraines. I wonder if there is such a thing as an antidote for MS?
My doctors have been thinking that I could not be correct but after giving them the facts they have swayed to our way of thinking.
My problem is that there seems to be little or nothing that can be done to shut down the headaches. I have found that I have Monoclonal Gammopathy which is an immune problem with possible severe side effects and wonder if maybe this has caused my immune system to act violently.
If you are so inclined, would you please respond to this missive.
Hi Ron, It sounds like there is a lot going as this has been a chronic issue for over 2 decades. Many factors can contribute to the development of migraines. I highly recommend that you work with a well trained functional health practitioner that can help you get to the root of this: https://drjockers.com/functional-nutrition-tips-to-find-a-great-health-coach/
I too, suffer a reaction to magnesium stearate. I believe it is more the magnesium. My back is set on fire and I get a horrible headache. I collected all my pills and a sample of stearate and 1 of magnesium and went off to the allergest. The headache roared and my back flared with the burning sensation, but no rash. The allergist said no allergy and left the room, never to return for questions. I changed everything i could to gel tabs, all but 3. I still have a headache everyday. When I try to eat foods with magnesium, I will get the same reaction. I know my body needs some, but I suffer when it reaches a certain limit.
Sorry to hear about this! I would try doing Epsom salt baths and see how you do. Blessings!
any plans on being able to ship to Canada
My daughter is on the autism spectrum and suffers from severe anxiety in addition to severe obsessive thoughts and compulsions that have worsened within the past two to three years. Would this magnesium supplement be helpful for her? Also, I must take magnesium for my heart and autoimmune conditions (rheumatoid arthritis, Feltys Syndrome, severe muscle cramps and diabetes). My cardiologist prescribed magnesium oxide, although I’ve read that this is one of the worst forms of magnesium. Should I change the form of magnesium I take? I find that I must take more magnesium in order to not have the severe muscle cramping I experience just from minor activities as grocery shopping over thirty minutes.
Sorry to hear about this! I would recommend the Brain Calm Magnesium for your daughter and the Mg-K Citrate for your muscle cramps
Brain Calm Magnesium: https://store.drjockers.com/products/brain-calm-magnesium
Mg-K Citrate: https://store.drjockers.com/products/mg-k-citrate
Hello. Infographics are useful! I have a question that no one has clear ans for!
Potassium sorbate, Sodium benzoate _have avoided these as much as possible for 2 decades! Yet, even premium, organic, Non-GMO products contain these. Oral supplements esp Liquid; & Skin care products and they say they are minor preservatives 🙁 I am unable to totally avoid..
Need some info on Cultured Dextrose and Yeast in bread (organic/non-GMO) items..
Yes definitely want to avoid the benzoates – those are very problematic!
You say it’s not bad, but I have experience that says different. I worked for a natural doc for a number of years, and one thing became evident. The ones with the stearate often times didn’t work. I had the tests to prove it. Plain and simple. Then I switched the patients to one with zero stearates and they worked. This happened over and over again. This is why I simply won’t use supplements with that hydrogenated oil.
Btw there are manufacturing equipment that works without it, but they cost more. Hence companies looking for every excuse to use them, because they have the old machines, amd don’t want to invest in the better ones. Don’t belive them doc,
I had gastric bypass surgery two years ago. Since then I feel foggy all the time. One exception, when I first wake up in the morning. No Doctor I have talked to seems to know why. Also eating is an issue. I feel so tired and sleepy when I eat. I definitely need help, do you recognize these systems and if I so which of your products do you recommend. The person that recommend you said that she took one of your products and passed a super long tape worm. Could that have anything to do with my symptoms? Please contact me at silva.benita@yahoo.com. Thank you for your help and advise…