How Do Phages Improve the Microbiome?
Phages are one of the newest discoveries in our exploration of the gut flora. The digestive tract is loaded with trillions of microorganisms that form a natural ecosystem commonly called the microbiome. Every day our body must maintain a certain balance of progenic and pathogenic organisms. The discovery of phages as a key component in our mucous membranes plays a critical role in stabilizing bacterial balance (1).
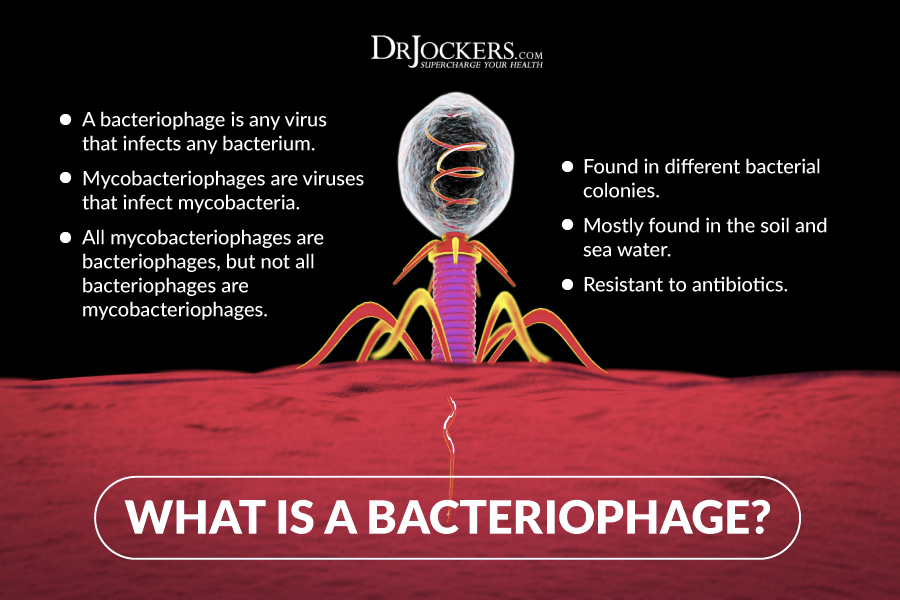
Bacteriophages which are also called “phages,” are viruses that infect and replicate within bacteria. Wherever bacteria and other microorganisms reside you will also find phages. Phages are one of the most abundant life forms on earth. They are found in water, soil, within, and on humans, plants, and animals. There are an estimated 1032 viral particles in existence.
Phages, like other viruses, have their genetic material enclosed in a protein sheath called a capsid, and are metabolically inert outside of their host bacterial cells. They are named based on certain characteristics that describe them. For example, siphoviridae are phages that have a non-enveloped head and non-contractile tail while myoviridae have a non-enveloped head and neck between their head and tail. Within these groups, there are hundreds of different subtypes.
The History of the Bacteriophages
Many traditions report stories of river waters having miraculous healing ability with infectious diseases. In fact, phages are found in abundance in free-flowing river waters and most scientists agree that phage count is extremely important to healthy spring water and keeping bacterial counts down.
In 1896, Ernest Hanbury Hankin reported in his journal that some sort of compound or organism in the Ganges and Yamuna rivers in India had powerful antibacterial action and prevented the spread of cholera (2). He observed that whatever it was able to pass through a porcelain filter.
British scientist Frederick Twort discovered a small agent that infected and killed bacteria in 1915. He hypothesized that it may be a sort of bacterial virus. His work was interrupted by the onset of World War I but in 1917, French-Canadian microbiologist Felix d’Herelle, found “an invisible, antagonistic microbe of the dysentery bacillus.”
D’Herelle, who was working at the Pasteur Institute in Paris at the time of discovery, knew he had made a breakthrough discovery. He reported, “In a flash, I had understood what caused my clear spots was in fact an invisible microbe…a virus parasitic on bacteria.” D’Herelle would be the first to call the virus a bacteriophage meaning “bacteria-eater” as phagein is the Greek meaning for “to eat.”
D’Herelle would go on to conduct much research into bacteriophages and develop the concept of phage therapy. This spread through Eastern Europe in areas of France and in particular Russia where they have been used for over 90 years as an alternative to antibiotics. Many experts believe they are perhaps our greatest weapon against MRSA and other drug resistant strains of bacteria (3, 4).
How Phages Interact with Bacterium
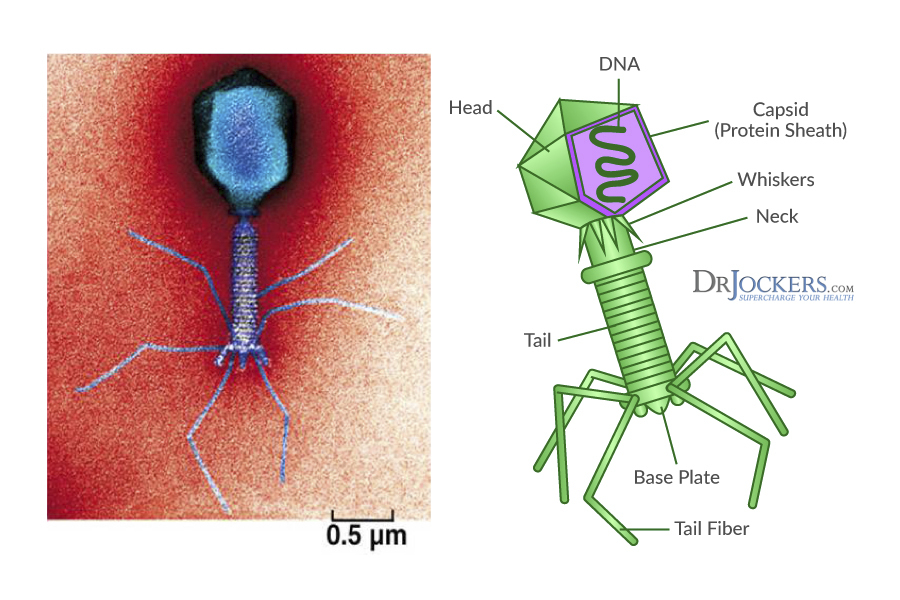
Phages have a specialized structure with tunnel tails that allow them to bind to the surface of their bacterial targets. Over 95% of phages in the world are caudovirals, meaning they inject their DNA into the bacterial cell through their tail. The hollow heads of the phage organisms hold their DNA and RNA and the goal of the phage is to inject this and breed through the use of the bacterial cell.
Once they are firmly attached the tail injects the viral DNA into the host cell. According to Phages.org once the phage has attached itself
“The viral DNA is then injected through the tail into the host cell, where it directs the production of progeny phages, often over a hundred in half an hour. These ‘young’ phages burst from the host cell (killing it) and infect more bacteria.”
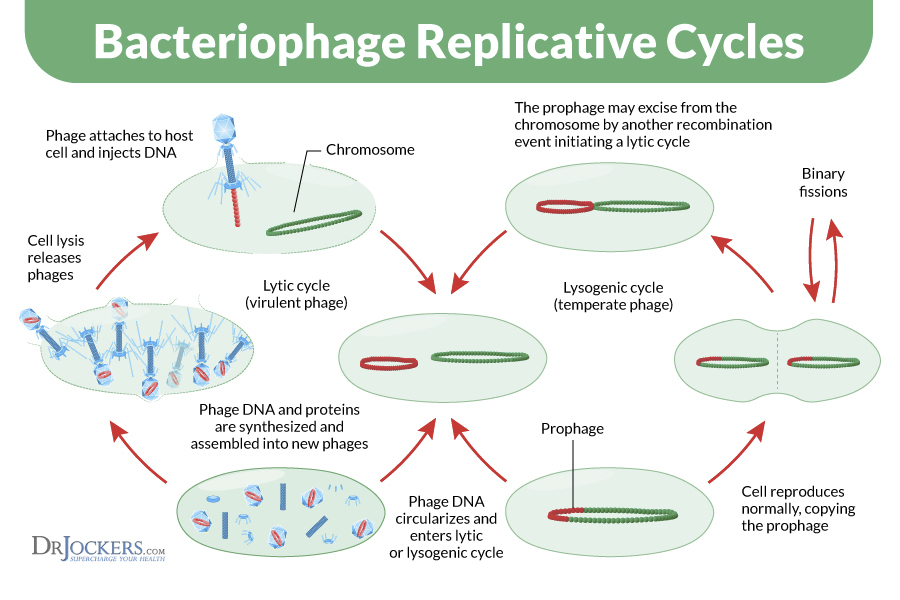
Phages have two major ways they affect bacterium. They have lysogenic and lytic activity. Lysogenic activity strengthens the bacterium by giving it genetic material that enhances environmental adaptation. Meanwhile, lytic activity destroys the infected bacterium by overwhelming it with progeny phages that burst through the host cell.
Phages are considered self-amplifying and self-limiting. A single dose of phage would continue to replicate and maintain a high concentration of phage particles as long as susceptible bacteria are present. They are also self-limiting because those phages that aren’t used are quickly flushed from the body by the immune system in the absence of host bacterial cells.
The Role of Mucous in the Body
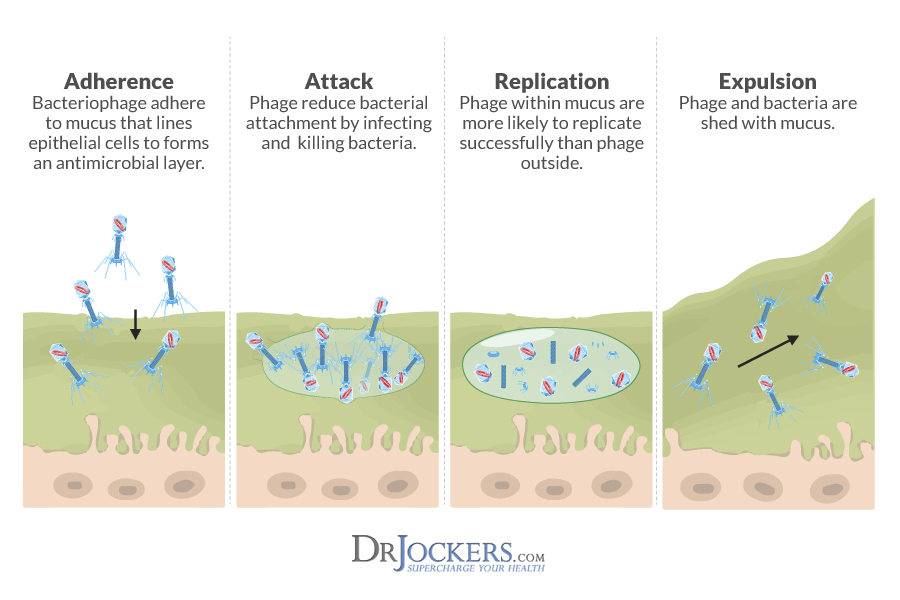
Mucosal surfaces are the primary entry points into the body for pathogenic microorganisms. Until recently most scientists viewed mucus as simply a physical barrier that helped to prevent the invasion of infectious organisms. It was also thought to play a lubricating role between tissues. However, the latest research shows that mucous appears to be the major home of phages.
The saliva around the human gums was found to have five phages to every one bacterium cell. On the mucosal surface of the gum itself, the ratio was close to forty-to-one.
Phages will be found wherever you find bacteria since they depend upon them for survival. Researchers have found evidence that phages partner with host animals and humans to kill off unwanted bacterial colonies and control the composition of friendly microorganisms in the body (5).
Sea Coral Slime and Human Mucus
Sea coral naturally produces and maintains a layer of surface slime which is similar to mucus. Researchers have noted that this slime contains massive amounts of phages and they protect the coral from certain bacterial species that would be dangerous to the coral reef. The coral slime is a sort of probiotic defense system in that it encourages beneficial organisms and protects against parasites and infectious species (6).
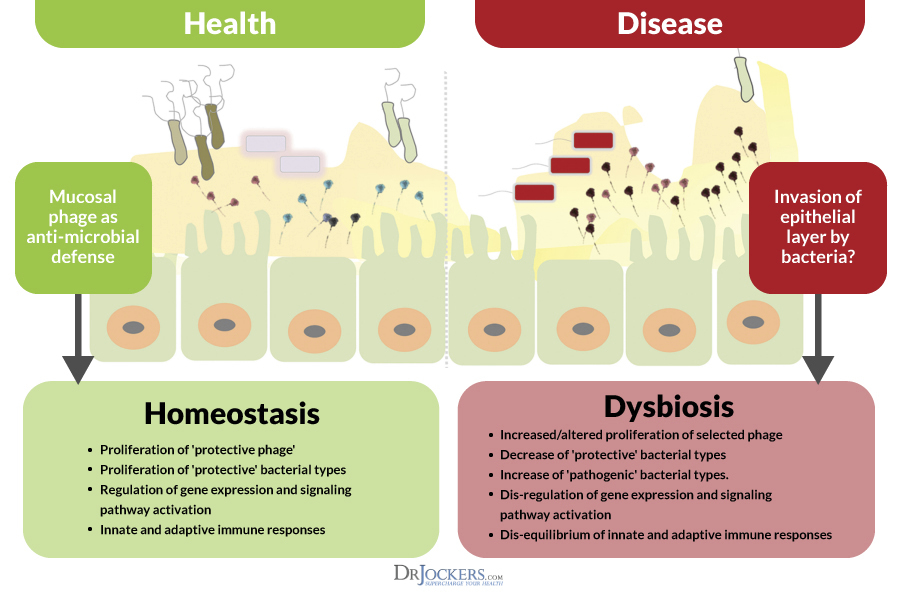
It is hypothesized that humans and other animals have adapted to select specific phages that give them a survival advantage against specific bacteria. It has been shown that sometimes phages insert genetic material into a friendly bacterium without killing it. This is called “prophagic lysogeny,” and offers the bacterium a survival advantage by improving its fitness and ability to defend against attack by other species of bacteriophages.
When the body is not under infectious stress the phage preferentially uses lysogenic activity to reproduce. This is non-threatening to the host bacterium. Under times of infectious stress, the phages favor a lytic function where they destroy the infected bacterium. It appears that communication between progenic bacterium, and the body’s immune system induces the bacteriophage to favor lysogenic or lytic activity.
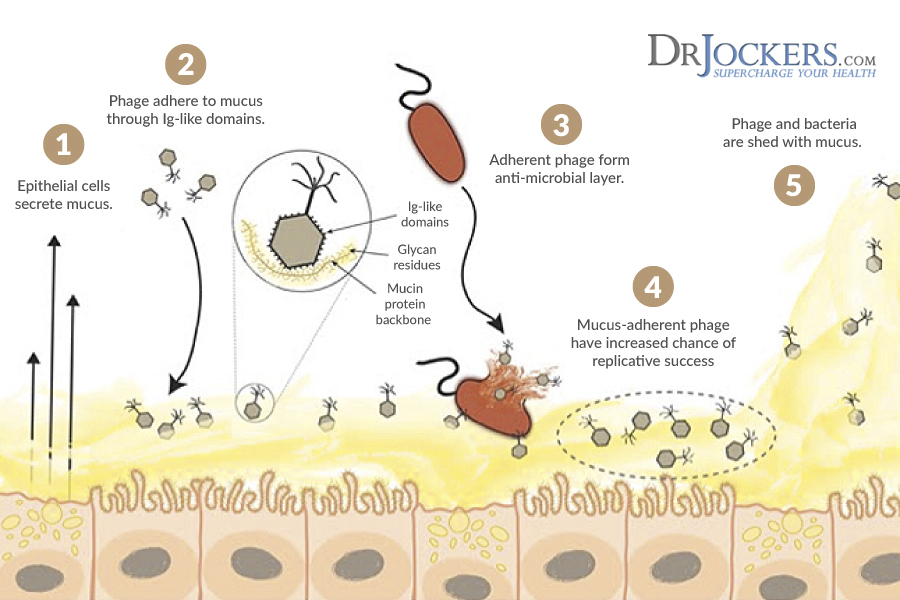
Mucosal Membrane Intelligence:
Researchers such as Jeremy Barr describe this as a form of mucosal intelligence system. Our microbial bacteria have been observed to create their own weapons in the form of phages to battle against opportunistic bacteria that may consume resources. This is a key part of the bacterial warfare that is going on within our bodies and plays an enormous role in our overall health.
Individuals with irritable bowel disorder (IBS), Crohn’s disease, and ulcerative colitis have a severely disrupted gut flora and a breakdown in the mucosal lining of their gut (7). These are indications that they are not receiving the vital protection that mucosal based phages offer and this must be considered as a major factor in the cause and progression of these disorders.
Phage Therapy and Anti-Biotics
In the 1920’s and 1930’s phage therapy was used for treating bacterial infections in the US. They were commonly used for soldiers in the Red Army. In the 1930’s, Stalin had his medics use phage therapy to help his troops ward off dysentery and it was heralded for its ability to help sick and wounded soldiers return to battle quickly.
Eli Lilly began to commercialize phage therapy in the US in the 1940’s. However, when antibiotics were produced in the 1950’s they were widely marketed and became the cornerstone of the pharmaceutical companies’ business. This put an end to research and development of phage therapy in the US and much of Europe.
The Soviet Union continued to use phage therapy and conducted many successful trials but their publications were mainly in Russian or Georgian languages and were unavailable internationally until recent years. The thorough research done over the last 80 years does indicate an 80-95% success rate with very minimal side effects.
In Russia and Eastern European countries such as Germany and Poland, it has been used successfully against diphtheria, tetanus, gangrene, scarlet fever, meningococcus, Salmonella, and Shigella. Hospitals in Eastern Russia region of Georgia spray operating rooms with phages before surgeries as prevention against infection. They are known to douse the air and surgical instruments with phages.
Phages Have Advantages over Anti-Biotics
Phage therapy has shown great promise for individuals suffering from MRSA, Sraphylococcus, Streptococcus, Pseudomonas, Salmonella, skin (acne) and soft tissue, gastrointestinal, respiratory, and orthopedic infections. Our society has an epidemic of anti-biotic resistance and the detrimental effects of antibiotic overuse which may be remedied by the advancement of phage therapy (8).
Phage therapy goes deeper than anti-biotics due to the phage’s ability to self-amplify when there is enough of the bacterial species they are targeting. Anti-biotics and probiotics for that matter are much more concentration dependent than phage. With phage the more of the targeted pathogenic species there are the more phage will target, replicate, and expand. Once the pathogenic species numbers are reduced the phage will naturally reduce and be swept out of the body.
Phage therapy has distinct advantages over antibiotics because the use of “wild type” phage with a multitude of different types of phages is too variable for bacterial resistance to form. Additionally, the phages keep in-tact healthy microflora and maintain a good microorganism count by preventing small intestinal bacterial overgrowth (SIBO).
The viruses do not affect or infect human tissue at all and once they are no longer able to find a host bacterial cell they are quickly neutralized by the immune system and/or swept out of the body through the feces. Phages ability to fend off opportunistic pathogenic organisms without any adverse reactions makes them a valuable health promoting tool.
Phages have been used in a variety of ways including systematically, topically, intravenously, and orally. Studies since 1966 have shown an 80-95% success rate with minimal side effects. They seem to be most effective against E. Coli, Acinetobacter, Psuedomonas, Staphylococcus aureus, and MRSA staph infections.

Limitations in Phage Therapy Research
Weld and other researchers have stated that up until recently phage biology was poorly understood and therefore the wrong phage and phage concentrations were used to treat infections. This was a limiting factor along with the poor quality of the phage preperations that were administered in many of the studies.
Current phage therapy research uses a “cocktail” consisting of a variety of phages that have the ability to target a number of different pathogenic bacteria including those with mutations or plasmids that protect them from other phages. Research using these phage cocktails over the next 5-10 years may very well revolutionize current treatment methods for infectious diseases as well as preventative measures.
Additionally, it seems that phage therapy may be combined with natural antimicrobial therapies for a unique synergistic approach to fighting bacteria in the body (9, 10).
Obstacles to Phage Therapy
The biggest obstacle to phage therapy is the relative lack of overall published research that would demand widespread usage. This is primarily due to low return on investment from pharmaceutical companies. Patented antibiotic therapies are a huge money maker for these companies whereas phage therapy is not expected to bring anywhere near the same returns.
The economic consequences on these large drug manufacturers dictate where they focus their time and energy. Globalyz Biotech is an international company that commercializes bacteriophage treatment.
Another major obstacle with phage therapy is the public’s negative view of viruses. There will take some education to cross the “psychological barrier” that people have so they will not be worried about using these live viruses as a healthful tool. We can look to Phages as the equivalent of probiotics and perhaps even title them as “proviriotics.”
We recently began carrying a specific anti-microbial supplement called Candida that includes the 4 most well-researched phages in it (Myovirdae T4, Myovirdae LH01, Myovirdae LL12, and Siphovirdae LL5). It also includes a host of powerful herbs and probiotics for an all in one anti-microbial formula.





